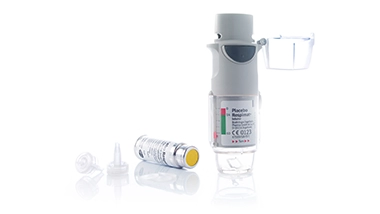
Medical packaging from cleanroom production in accordance with DIN EN ISO 13485
AWe develop and manufacture high-quality primary and secondary packaging for medical products as well as functionally relevant components and assemblies for medical devices. Our GMP-compliant manufacturing is DIN EN ISO 13485 certified and based on fully automated cleanroom production in accordance with DIN EN ISO Class 7 (GMP Standard C), which enables maximum product safety and hygiene. From dental packaging and medical connectors to complex injection-molded parts for medical technology packaging, we accompany you from the concept phase through validation to series production. Thanks to our many years of experience in medical packaging, we combine precision with efficient manufacturing technology, also as a contract manufacturer. Our in-house toolmaking department enables individual adaptations and fast implementation.

Baby flow mask
ventilation mask for infants

Inhalation mask
for children

Filter housing
with 2K lid

dental cartridges
manufactured using precision injection molding
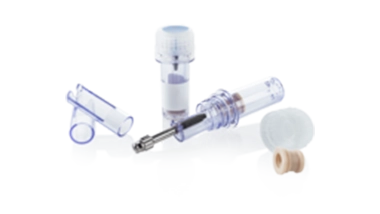
Implant packaging
for dental applications
Primary and secondary packaging of the highest quality:
GMP-compliant, ISO 13485 certified, and manufactured in a fully automated cleanroom
Our expertise in medical packaging includes sophisticated plastic components such as injection-molded packaging, ventilation masks, and dental applications. Manufacturing takes place in a fully automated cleanroom production facility in accordance with DIN EN ISO Class 7 (GMP Standard C) with maximum product safety for medical products and medical devices. From prototype development and validation to series production, we also support complex projects and innovative new developments. State-of-the-art injection molding equipment, precise assembly technology, and our in-house toolmaking facility enable us to provide customized packaging solutions that are flexible, efficient, and certified according to DIN EN ISO 13485. Thanks to our cleanroom technology with end-to-end automation, we set standards in precision, GMP compliance, and manufacturing quality without compromise.
Our Added Value,
Your Benefits.

MICHAEL
Your BENEFIT: Everything from a single source.
- Customized development of primary and secondary packaging for medical devices – tailor-made, DIN EN ISO 13485 certified, and GMP compliant
- Contract manufacturing for pharmaceutical packaging and medical devices – from the concept phase to series production
- Fully automated cleanroom in accordance with DIN EN ISO Class 7 / GMP Standard C – less particle sources, even greater safety
- Documentation in accordance with GMP (Good Manufacturing Practice) rules – certified in accordance with DIN EN ISO 13485
- Tool procurement via our in-house toolmaking department
We would be happy to present our advantages to you in a personal meeting.
INDIVIDUAL PRODUCT DEVELOPMENT
FOR MEDICAL PACKAGING AND MEDICAL DEVICES
We develop and produce high-quality components and assemblies for medical packaging and medical devices, tailored to your specific requirements. Our product development combines state-of-the-art manufacturing technology with fully automated cleanroom production in accordance with DIN EN ISO Class 7 (GMP Standard C) to enable maximum application safety and the highest level of product protection. Seamless quality management in accordance with DIN EN ISO 13485 accompanies every project – from the concept phase through prototyping and validation to series production. We focus on customer-specific packaging solutions that combine technical precision with economic efficiency.
PÖPPELMANN FAMAC®.
Awarded and certified:

Systematic quality management for medical devices

International Sustainability &
Carbon Certification

Systematic
energy management

Systematic
environmental management

Management system
for health and safety